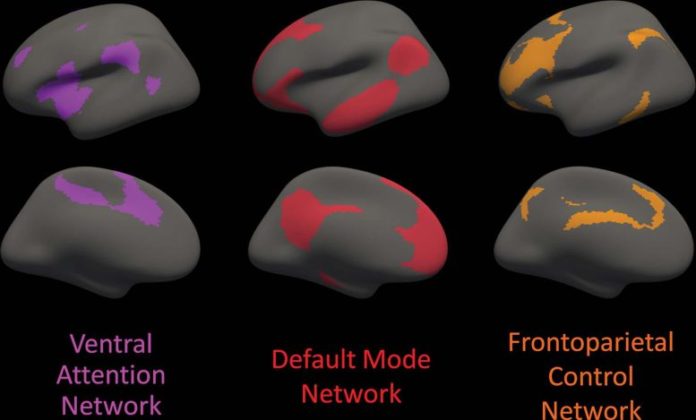
Cortical locations included with each of the 3 dispersed cortical association networks evaluated are shown on an inflated brain surface area, seen from the lateral element (leading row) and the median element (bottom row) of the left hemisphere; the ideal hemisphere (disappointed) is almost a mirror image of the left hemisphere. The areas are based upon a basic practical parcellation of the human brain from a resting-state practical MRI research study of 1,000 healthy people. The 3 particular networks of interest are the forward attention network (purple), default mode network (red), and frontoparietal control network (orange). Credit: Radiological Society of North America
The structural stability of the brain’s white matter as determined with a sophisticated MRI strategy is lower in cognitively typical individuals who bring a hereditary anomaly related to Alzheimer’s illness than it remains in non-carriers, according to a research study in Radiology Researchers stated the findings reveal the guarantee of commonly offered imaging strategies in assisting to comprehend early structural modifications in the brain prior to signs of dementia emerge.
People who bring the autosomal dominant Alzheimer illness (ADAD) anomaly have a greater danger of Alzheimer’s illness, a kind of dementia that impacts about one in 9 individuals in the UnitedStates The anomaly is connected to an accumulation of irregular protein called amyloid-beta in the brain that impacts both the noodle and the signal-carrying white matter.
“It’s thought that the amyloid deposition in the gray matter could disrupt its function, and as a result the white matter won’t function correctly or could even atrophy,” stated research study lead author Jeffrey W. Prescott, M.D.,Ph D., neuroradiologist at the MetroHealth Medical Center in Cleveland.
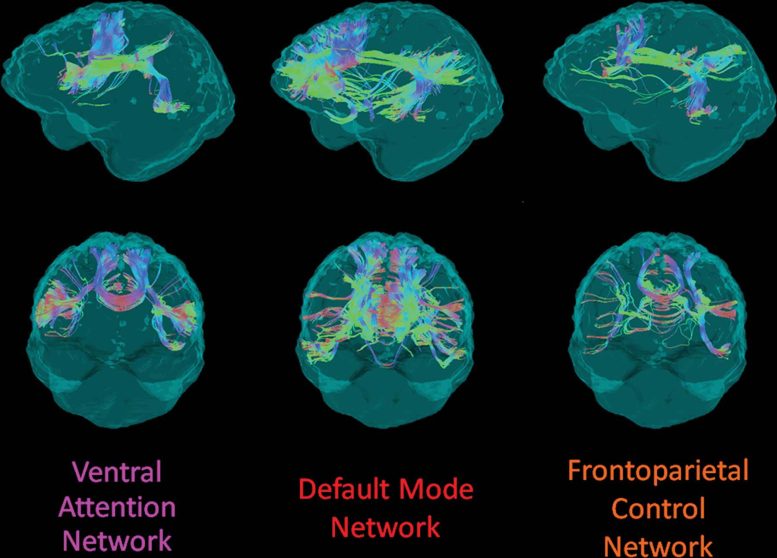
Example structural connection of the 3 evaluated dispersed cortical networks from one individual in the DIAN accomplice, saw laterally from the left (leading row) and anteriorly (bottom row). Streamline colors show directionality of water diffusion at diffusion tensor imaging: green = anteroposterior, red = left-right, blue = superoinferior. Credit: Radiological Society of North America
An earlier research study byDr Prescott and coworkers on clients with erratic Alzheimer’s illness, which makes up 99% of cases, discovered that white matter structural connection, as determined with an MRI strategy called diffusion tensor imaging (DTI), deteriorated substantially as clients established more amyloid problem.
“The current work extends these results by showing that similar findings are detectable in asymptomatic at-risk patients,” stated Jeffrey R. Petrella, M.D., teacher of radiology at Duke University and senior author on both research studies.
In the brand-new research study,Dr Prescott and coworkers utilized information from the Dominantly Inherited Alzheimer Network (DIAN) to compare ADAD anomaly providers with non-carriers to see if there were modifications in structural connection that might be associated with the anomaly.
The research study individuals consisted of 30 anomaly providers, suggest age 34 years, and 38 non-carriers, suggest age37 The individuals all had typical cognition when they went through structural brain MRI and DTI.
Analysis revealed that anomaly providers had lower structural connection in the frontoparietal control network, which links locations primarily in the parietal and frontal lobes, 2 areas understood to be included with Alzheimer’s illness. Among anomaly providers, there was a connection in between anticipated years up until start of signs and white matter structural connection in the frontoparietal control network, even when managing for amyloid plaque problem.
“This suggests that DTI measures of network integrity may serve as a surrogate for the brain’s resilience to pathologic attack,”Dr Petrella stated.
“We used a network measurement called global efficiency, in which a decreased efficiency can be taken as a breakdown in the organization of the network,”Dr Prescott included. “The results show that for mutation carriers, global efficiency would decrease significantly as they approach the estimated age of symptom onset.”
The research study findings support a prospective function for imaging-based recognition of structural modifications of the brain in individuals at hereditary danger for early-onset Alzheimer’s illness in comprehending how genes affect the illness procedure that results in dementia.
“This shows the potential of MRI as an evaluation tool in patients who are deemed at-risk for Alzheimer’s disease before they develop symptoms,”Dr Prescott stated. “Use of these advanced MRI techniques could help further refine identification of at-risk patients and risk measurements.”
The findings likewise indicate a function for imaging in studying restorative drugs to deal with Alzheimer’s illness. While most of trials up until now have actually been carried out with clients who currently have Alzheimer’s illness or cognitive disability, earlier recognition and treatment of clients at danger represents a more appealing opportunity for avoiding or a minimum of postponing the start of dementia.
“One potential clinical use of this study tool would be to add quantitative information to risk factors like family history and use that to help identify patients early, when they may benefit from treatment,”Dr Prescott stated. “But until we have an effective treatment, we will have to wait for that to be implemented.”
The scientists want to do a follow-up utilizing sophisticated imaging and upgraded information from the DIAN network to assess the development of Alzheimer’s illness in the research study individuals.
Reference: “Diffusion Tensor MRI Structural Connectivity and PET Amyloid Burden in Preclinical Autosomal Dominant Alzheimer Disease: The DIAN Cohort” by Jeffrey W. Prescott, P. Murali Doraiswamy, Dragan Gamberger, Tammie Benzinger, Jeffrey R. Petrella, for the Dominantly Inherited Alzheimer Network, 12 October 2021, Radiology
DOI: 10.1148/ radiol.2021210383
Collaborating withDrs Prescott and Petrella were P. Murali Doraiswamy, M.B.B.S., Dragan Gamberger,Ph D., and Tammie Benzinger, M.D.,Ph D., for the Dominantly Inherited Alzheimer Network.