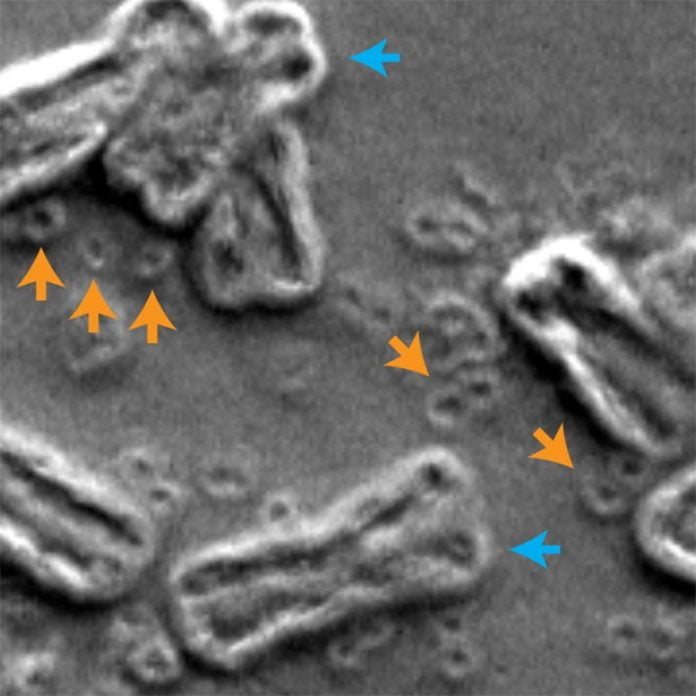
In this scanning electron micrograph of inside the nucleus of a cancer cell, chromosomes are shown by blue arrows and circular extra-chromosomal DNA are shown by orange arrows. Credit: Image thanks to Paul Mischel, UC San Diego
Researchers discover that the phenomenon of chromothripsis leads to reorganized genomes and extra-chromosomal DNA that assists altered cells not just avert treatment, however end up being more aggressive.
Cancer is among the world’s biggest health conditions since, unlike some illness, it is a moving target, continuously developing to avert and withstand treatment.
In a paper released in the December 23, 2020, online concern of Nature, scientists at University of California San Diego School of Medicine and the UC San Diego branch of the Ludwig Institute for Cancer Research, with associates in New York and the United Kingdom, explain how a phenomenon referred to as “chromothripsis” separates chromosomes, which then reassemble in manner ins which eventually promote cancer cell development.
Chromothripsis is a disastrous mutational occasion in a cell’s history that includes huge rearrangement of its genome, instead of a progressive acquisition of rearrangements and anomalies in time. Genomic rearrangement is a crucial quality of numerous cancers, permitting altered cells to grow or grow quicker, untouched by anti-cancer treatments.
“These rearrangements can occur in a single step,” stated very first author Ofer Shoshani, PhD, a postdoctoral fellow in the laboratory of the paper’s co-senior author Don Cleveland, PhD, teacher of medication, neurosciences and cellular and molecular medication at UC San Diego School of Medicine.
“During chromothripsis, a chromosome in a cell is shattered into many pieces, hundreds in some cases, followed by reassembly in a shuffled order. Some pieces get lost while others persist as extra-chromosomal DNA (ecDNA). Some of these ecDNA elements promote cancer cell growth and form minute-sized chromosomes called ‘double minutes.’”
Research released in 2015 by researchers at the UC San Diego branch of the Ludwig Institute for Cancer Research discovered that approximately half of all cancer cells in numerous kinds of cancers consist of ecDNA bring cancer-promoting genes.
In the most recent research study, Cleveland, Shoshani and associates utilized direct visualization of chromosome structure to determine the actions in gene amplification and the system underlying resistance to methotrexate, among the earliest chemotherapy drugs and still commonly utilized.
In partnership with co-senior author Peter J. Campbell, PhD, head of cancer, aging and somatic anomaly at Wellcome Sanger Institute in the United Kingdom, the group sequenced the whole genomes of cells establishing drug resistance, exposing that chromosome shattering jump-starts development of ecDNA-carrying genes that give anti-cancer treatment resistance.
The researchers likewise determined how chromothripsis drives ecDNA development after gene amplification inside a chromosome.
“Chromothripsis converts intra-chromosomal amplifications (internal) into extra-chromosomal (external) amplifications and that amplified ecDNA can then reintegrate into chromosomal locations in response to DNA damage from chemotherapy or radiotherapy,” stated Shoshani. “The new work highlights the role of chromothripsis at all critical stages in the life cycle of amplified DNA in cancer cells, explaining how cancer cells can become more aggressive or drug-resistant.”
Said Cleveland: “Our identifications of repetitive DNA shattering as a driver of anti-cancer drug resistance and of DNA repair pathways necessary for reassembling the shattered chromosomal pieces has enabled rational design of combination drug therapies to prevent development of drug resistance in cancer patients, thereby improving their outcome.”
The findings attend to among the so-called 9 Grand Challenges for cancer treatment advancement, a joint collaboration in between the National Cancer Institute in the United States and Cancer Research UK, the world’s biggest independent cancer research study and awareness charity.
Reference: “Chromothripsis drives the evolution of gene amplification in cancer” by Ofer Shoshani, Simon F. Brunner, Rona Yaeger, Peter Ly, Yael Nechemia-Arbely, Dong Hyun Kim, Rongxin Fang, Guillaume A. Castillon, Miao Yu, Julia S. Z. Li, Ying Sun, Mark H. Ellisman, Bing Ren, Peter J. Campbell and Don W. Cleveland, 23 December 2020, Nature.
DOI: 10.1038/s41586-020-03064-z
Co-authors consist of: Ofer Shoshani, Peter Ly, Yael Nechemia-Arbely, Dong Hyun Kim, Rongxin Fang, Miao Yu, Julia S. Z. Li, Guillaume A. Castillon, Ying Sun, Mark H. Ellisman and Bing Ren, all at UC San Diego; Simon F. Brunner, Wellcome Sanger Institute; and Rona Yaeger, Memorial Sloan Kettering Cancer Institute, NY.
Funding for this research study came, in part, from the National Institutes of Health (grants R35 GM122476 and K99 CA218871), the Wellcome Trust, the Swiss National Science Foundation, the Ludwig Institute for Cancer Research, the Memorial Sloan Kettering Cancer Center, the National Institute of General Medical Sciences (P41GM103412, R24GM137200) and the High End Instrumentation Award.