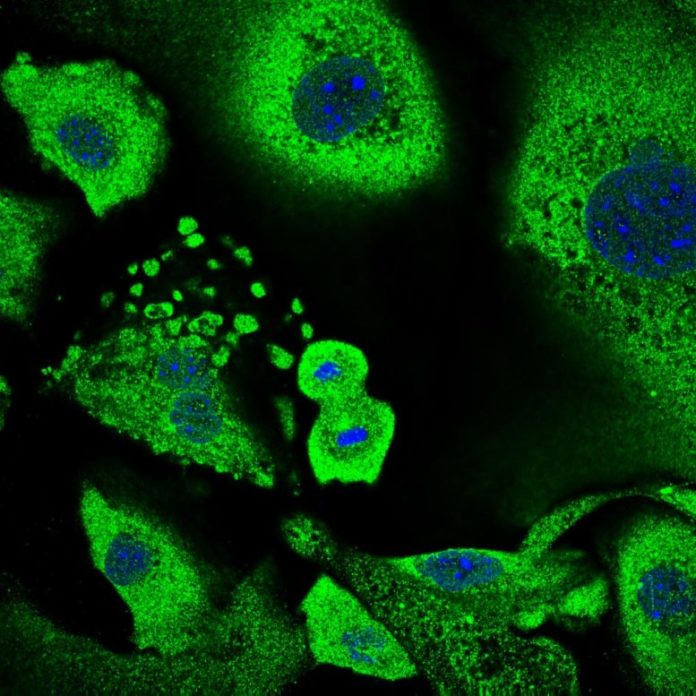
A keratinocyte going through cellular division surrounded by senescent keratinocytes. The total size of the cells and their nucleus are common qualities of senescence, which play a crucial function in growth suppression. Credit: Annagiulia Ciocia/ CRG
New research study discovers molecular systems that collaborate skin cell senescence.
A research study released today (January 11, 2022) in Cell Reports exposes crucial insights into the molecular systems that underpin the body’s natural defenses versus the advancement of skin cancer. The findings provide brand-new hints into the habits of skin cancer at the cellular level, leading the way for possible brand-new healing targets to deal with the illness.
“We found that the protein CSDE1 coordinates a complex chain of events that enable senescence in skin cells, significantly slowing down their function without causing death,” states Rosario Avolio, very first author of the research study and postdoctoral scientist at the CRG at the time of submission. “The resulting cells act as a firewall against cancer, suppressing the formation of tumors.”
Researchers led by Fátima Gebauer at the Centre for Genomic Regulation (CRG) performed the research study by gathering keratinocytes from mice, the most plentiful kind of skin cell in the skin. Keratinocytes can trigger numerous kinds of skin cancer consisting of basal and squamous cell cancers, 2 of the most often taking place types of all human cancers.
The group experimentally presented genes that drive the development of cancer, which caused the cells into a state of senescence. They discovered that when levels of CSDE1 were diminished, cells might not go through senescence and ended up being celebrated, a needed action in the advancement of cancer.
Further experiments exposed that when CSDE1-depleted cells were implanted under the skin of mice, they began forming deadly growths. The authors discovered this striking due to the fact that every dealt with mouse established squamous cell cancers within 15 to 20 days, highlighting the value of CSDE1 in growth suppression.
The scientists found that CSDE1 promotes growth suppression through 2 various systems. CSDE1 causes the cell to produce a mixed drink of cytokines and enzymes that require the cell to go into a state of irreversible development arrest. CSDE1 likewise stops the synthesis of YBX1, a protein formerly understood to promote tumor development and aggressiveness.
According to the authors, the findings of the research study are unexpected due to the fact that CSDE1 has actually been formerly connected to driving the development of cancers, not reducing them. Previous research studies from the very same group led byDr Gebauer discovered that CSDE1 promotes the development of metastases in cancer malignancy, a less typical however the most aggressive kind of skin cancer. Other research studies have actually revealed CSDE1 is connected to growth expansion in numerous kinds of cancer.
“CSDE1 is very much the ‘Dr. Jekyll and Mr. Hyde’ of proteins. It has an unpredictable dual nature depending on what type of cell and tissue it’s found in,” discussesDr Gebauer, acting co-coordinator of the Gene Regulation, Stem Cells and Cancer research study program at the CRG and senior author of the research study. “We do not know why this protein causes cancer in some cases and suppresses them in others. Exploring the root cause of this will have important implications for the discovery of new, more personalized cancer treatments.”
CSDE1 is an RNA– binding protein, a kind of protein that keeps track of RNA, frequently as quickly as they’re made with the possible to considerably alter their function. One possible theory that discusses why CSDE1 acts in a different way is that regular skin cells or growths each have somewhat various variations of the protein which impact the larger molecular equipment in various methods.
The research study is among the couple of to analyze the function of RNA-binding proteins in developing cell senescence, which is a crucial brand-new frontier in cancer research study. “It was long thought that RNA-binding proteins are universal molecules that cells use for general housekeeping, and that they cannot be targeted therapeutically. It is becoming increasingly clear that this is not true, and that this emerging field is critical for understanding human disease,” concludesDr Gebauer.
Reference: “Coordinated post-transcriptional control of oncogene-induced senescence by UNR/CSDE1” 11 January 2022, Cell Reports
DOI: 10.1016/ j.celrep.2021110211