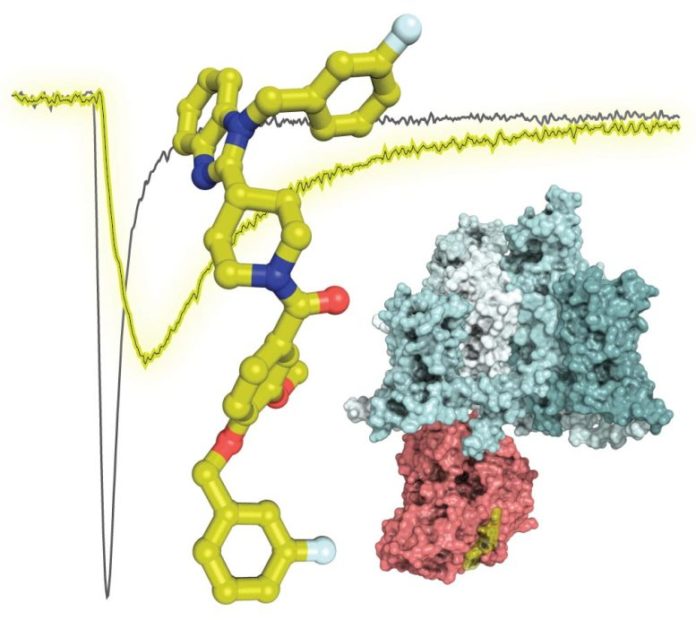
This image reveals the structure of substance 194 (yellow sticks) overlaid on an electrophysiology trace revealing lowered salt currents (yellow vs. black lines) from cells that are treated with substance194 The structure highlights the forecasted interaction of CRMP2 (pink, PDB 2GSE) with the Nav1.7 channel (cyan, PDB 6J8G). Credit: University of Arizona Health Sciences
Researchers targeted a typical salt ion channel to reverse discomfort, with favorable outcomes that might result in a non-addictive option to deal with discomfort.
Researchers at the University of Arizona Health Sciences are better to establishing a safe and efficient non-opioid painkiller after a research study revealed that a brand-new substance they produced decreases the feeling of discomfort by managing a biological channel connected to discomfort.
Most individuals experience discomfort at some time in their lives, and the National Institutes of Health quotes 100 million individuals in the U.S. struggle with persistent discomfort. Approximately 21-29% of clients recommended opioids for persistent discomfort abuse them and 8-12% of individuals utilizing an opioid for persistent discomfort establish an opioid usage condition, according to the National Institute on DrugAbuse In 2019, almost 50,000 individuals in the U.S. passed away from opioid-involved overdoses.
“Drug discovery for persistent discomfort is at the leading edge of this research study, and it’s being magnified by the crossway of the COVID-19 pandemic and the opioid epidemic,” stated Rajesh Khanna, PhD, associate director of the UArizona Health Sciences Comprehensive Pain and Addiction Center and teacher of pharmacology in the UArizona College of Medicine–Tucson “Drug discovery is a very arduous process. Our lab looked at a fundamental mechanism of pain, came up with a way to differentiate it from those before us and found a compound that has the potential as a new non-opioid treatment for pain.”

Rajesh Khanna, PhD, is the associate director of the University of Arizona Health Sciences Comprehensive Pain and Addiction Center and a teacher of pharmacology in the UArizona College of Medicine–Tucson Credit: University of Arizona Health Sciences/Kris Hanning
The paper, “Selective targeting of NaV1.7 via inhibition of the CRMP2-Ubc9 interaction reduces pain in rodents,” was released today (November 10, 2021) in Science Translational Medicine
The biological system at the heart of the research study is NaV1.7, a salt ion channel that formerly was connected to the feeling of discomfort through hereditary research studies of individuals with unusual discomfort conditions.
Nerve cells, or nerve cells, utilize electrical currents to send out signals to the brain and throughout the body, and salt ion channels are crucial to a cell’s capability to create those electrical currents. When a nerve cell is promoted, the NaV1.7 channel opens and enables favorably charged salt ions to cross the cell membrane and get in the formerly adversely charged cell. The modification in charge throughout the cell membrane creates an electrical present, which increases the excitability of the nerve cell and sets in movement a waterfall of occasions that results in discomfort.
Because NaV1.7 is a human-validated target for discomfort, numerous efforts have actually attempted to stop discomfort by utilizing salt ion channel inhibitors to obstruct NaV1.7. None have actually succeeded.Dr Khanna and his group took a various method– instead of obstruct NaV1.7, they wished to indirectly control it.
Using a substance they developed and called 194, the group effectively controlled NaV1.7 activation in the lab utilizing afferent neuron from 4 various types, consisting of human beings. In animal designs, 194 worked in reversing discomfort in 6 various discomfort designs in both sexes.
Researchers likewise discovered that 194 likewise might promote discomfort relief by triggering the body’s endogenous, or naturally taking place, opioid system. Once produced, endogenous opioids trigger receptors that produce physiological modifications such as discomfort relief. And 194 did so without triggering motor efficiency concerns, depressive habits or dependency.
Finally,Dr Khanna and the group observed a synergistic result when 194 was integrated with morphine and gabapentin. This is an appealing indication that 194 might likewise be utilized in a dose-reduction method for pain relievers that have unfavorable negative effects, consisting of opioids, while preserving high levels of discomfort relief.
The science behind 194
Dr Khanna’s previous research study determined a protein, collapsin action conciliator protein 2 (CRMP2), and an enzyme, Ubc9, that both contribute in NaV1.7 activation. CRMP2 is a protein that binds to NaV1.7 and carries it to the cell membrane, where salt ions are then moved into the cell. Ubc9 is an enzyme that tags CRMP2 with another protein– a little ubiquitin-like modifier protein– to particularly direct control of NaV1.7.
Building on this understanding,Dr Khanna and the group set out to figure out if they might straight control the activity of NaV1.7 by obstructing Ubc9 from engaging with CRMP2. Team members consisting of May Khanna, PhD, associate teacher of pharmacology and BIO5 Institute member, Vijay Gokhale, PhD, associate research study teacher in the BIO5 Institute, and Samantha Perez-Miller, PhD, scientist and researcher in the Department of Pharmacology, taken a look at 50,000 existing little particles to recognize the ones with a structure comparable to Ubc9.
They chosen less than 50 of the closest matches, which were then evaluated inDr Khanna’s lab to see if their existence would reduce the increase of salt through NaV1.7. The findings were appealing, so the group set their sights on establishing a special, more efficient substance.
The result was 194, which UArizona trademarked and certified to start-up Regulonix LLC through Tech Launch Arizona, the UArizona workplace that advertises developments coming from university research study.Drs Khanna and Gokhale established Regulonix LLC in 2016 to attend to the growing opioid epidemic by establishing brand-new, non-addictive methods to deal with discomfort and advertising those developments.
While 194 reveals excellent pledge for discomfort relief,Dr Khanna and the group have actually been dealing with the National Institutes of Health’s National Center for Advancing Translational Sciences to enhance the substance. In this case, an NCATS group is mostly concentrating on enhancing 194’s half-life– the time it considers a drug to decrease by half in your body– and its drug-like homes.
It is a crucial action in enhancing the substance’s capacity as a pain-relieving drug and advancing to the next phase, where scientists will declare Food and Drug Administration approval to start medical trials.
Reference: “Selective targeting of NaV1.7 via inhibition of the CRMP2-Ubc9 interaction reduces pain in rodents” 10 November 2021, Science Translational Medicine