Proposed structure of Bi-bound zinc-binding domain of SARS-CoV-2 helicase. Through tossing out the essential zinc(II) ions in the zinc-binding domain of SARS-CoV-2 helicase, RBC showed its capability to potently reduce the duplication of SARS-CoV-2. Credit: The University of Hong Kong
HKU researchers and microbiologists collectively find an unique antiviral technique for treatment of COVID-19 utilizing existing metallodrugs.
A research study group led by Professor Hongzhe SUN, Norman & Cecilia Yip Professor in Bioinorganic Chemistry, Department of Chemistry, Faculty of Science, and Professor Kwok Yung YUEN, Henry Fok Professor in Infectious Diseases, Department of Microbiology, Li Ka Shing Faculty of Medicine of the University of Hong Kong (HKU), has actually found an unique antiviral technique for treatment of COVID-19.
They found that a class of metallodrugs presently utilized in the treatment of other transmittable illness is revealing effectiveness to potently reduce SARS-CoV-2 duplication and alleviate viral-associated signs in an animal design.
The findings supply a brand-new and easily offered restorative choice with high medical capacity for infection with SARS-CoV-2. This ground-breaking work has actually been released online in a top-class clinical journal Nature Microbiology. An associated patent has actually been submitted in the United States.

From the right: Dr Jasper F W CHAN and Dr Shuofeng YUAN of the Department of Microbioloty, Professor Hongzhe SUN and Dr Runming WANG of the Department of Chemistry. Credit: The University of Hong Kong
Background
SARS-CoV-2 is an emerging coronavirus that has actually triggered over 30 million laboratory-confirmed cases and more than 1 million deaths internationally of COVID-19 because December 2019. As the procedure of establishing an efficient vaccine is still continuous, another technique for avoidance and treatment of the illness is to recognize anti-COVID-19 representatives from existing virus-specific antiviral drugs to repurpose their usages to target the brand-new infection. Remdesivir, a broad-spectrum antiviral drug, has actually been reported to reveal effectiveness towards SARS-CoV-2. However, international scarcity of the drug, its reasonably high cost and absence of considerable medical advantages in extreme cases, are elements that have actually restricted its larger applications. Clinical trials on a series of antiviral representatives are still continuous which have yet to show restorative effectiveness. Therefore, higher efforts are required to extend the examination to cover a broader spectrum of scientifically authorized drugs, which ideally might break the ice to alternative treatment methods versus the illness through some easily offered channels.
Study approach and findings
Generally, metal substances are utilized as anti-microbial representatives; their antiviral activities have actually hardly ever been checked out. After evaluating a series of metallodrugs and associated substances, the research study group recognized ranitidine bismuth citrate (RBC), a typically utilized anti-ulcer drug which includes the metal Bismuth for treatment of Helicobacter pylori-associated infection, as a powerful anti-SARS-CoV-2 representative, both in vitro and in vivo.
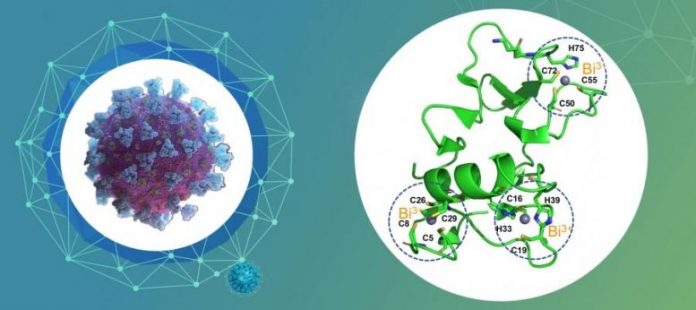
RBC targets the crucial non-structural protein 13 (Nsp13), a viral helicase necessary for SARS-CoV-2 to reproduce, by displacing the essential zinc(II) ions in the zinc-binding with Bismuth-ions, to potently reduce the activity of the helicase.
RBC has actually been shown to significantly decrease viral loads by over 1,000-folds in SARS-CoV-2-infected cells. In specific, in a golden Syrian hamster design, RBC reduces SARS-CoV-2 duplications to decrease viral loads by ~100 folds in both the upper and lower breathing systems, and reduces virus-associated pneumonia. RBC extremely lessens the level of prognostic markers and other significant pro-inflammatory cytokines and chemokines in extreme COVID-19 cases of contaminated hamsters, compared to the Remdesivir-dealt with group and control group.
RBC displays a low cytotoxicity with a high selectivity index at 975 (the bigger the number the more secure the drug), as compared to Remdesivir which has a low selectivity index at 129. The finding suggests a broad window in between the drug’s cytotoxicity and antiviral activity, which enables a fantastic versatility in changing its does for treatment.
The group examined the systems of RBC on SARS-CoV-2 and exposed for the very first time the crucial Nsp13 helicase as a druggable target by RBC. It irreversibly tosses out the essential zinc(II) ions in the zinc-binding domain to alter it to bismuth-bound through an unique metal displacement path. RBC and its Bi(III) substances defunctionalized the Nsp13 helicase and potently prevented both the ATPase (IC50=0.69 µM) and DNA-relaxing (IC50=0.70 µM) activities of this enzyme.
The research study findings highlight viral helicases as a druggable target, and the high medical capacity of bismuth(III) drugs and other metallodrugs for treatment of SARS-CoV-2 infections. Hopefully, following this essential development, more antiviral representatives from easily offered scientifically authorized drugs might be recognized for prospective treatment of COVID-19 infections. They can be in the kind of mix routines (mixed drinks) with drugs that show anti-SARS-CoV-2 activities consisting of RBC, dexamethasone and interferon-β1b.
Reference: “Metallodrug ranitidine bismuth citrate suppresses SARS-CoV-2 replication and relieves virus-associated pneumonia in Syrian hamsters” by Shuofeng Yuan, Runming Wang, Jasper Fuk-Woo Chan, Anna Jinxia Zhang, Tianfan Cheng, Kenn Ka-Heng Chik, Zi-Wei Ye, Suyu Wang, Andrew Chak-Yiu Lee, Lijian Jin, Hongyan Li, Dong-Yan Jin, Kwok-Yung Yuen and Hongzhe Sun, 7 October 2020, Nature Microbiology.
DOI: 10.1038/s41564-020-00802-x
Key members of the research study group:
Department of Chemistry, Faculty of Science
- Professor Hongzhe SUN, Norman & Cecilia Yip Professor in Bioinorganic Chemistry, Chair Professor of Chemistry
- Dr. Runming WANG, Post-doctoral Fellow, Department of Chemistry
Department of Microbiology, Li Ka Shing Faculty of Medicine
- Professor Kwok-Yung YUEN, Henry Fok Professor in Infectious Diseases; likewise a Co-Director of the State Key Laboratory of Emerging Infectious Diseases and Academician of the Chinese Academy of Engineering
- Dr. Shuofeng YUAN, Research Assistant Professor
- Dr. Jasper F W CHAN, Clinical Assistant Professor
Staunch Supporters
The work was supported by the Research Grants Council (RGC) of Hong Kong, the National Key R&D Programmes of China, the University of Hong Kong (HKU) Seed Fund for Basic Research and contributions from the Lo Ying Shek Chi Wai Foundation, Richard Yu and Carol Yu, the Shaw Foundation of Hong Kong, Michael Seak-Kan Tong, May Tam Mak Mei Yin, Hong Kong Sanatorium & Hospital, Hui Ming, Hui Hoy and Chow Sin Lan Charity Fund Limited, Chan Yin Chuen Memorial Charitable Foundation, Marina Man-Wai Lee, the Hong Kong Hainan Commercial Association South China Microbiology Research Fund, the Jessie & George Ho Charitable Foundation, Perfect Shape Medical Limited, Kai Chong Tong, Foo Oi Foundation Limited, Tse Kam Ming Laurence, and Norman & Cecilia Yip Foundation.