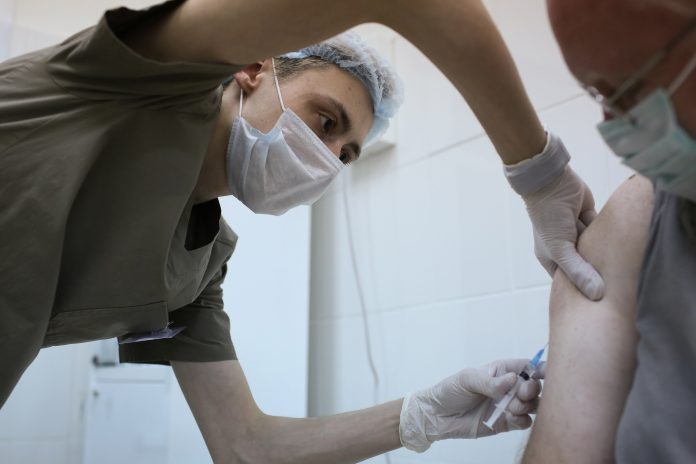
A heath employee injects the ‘Gam-COVID-Vac’, likewise referred to as ‘Sputnik V’, Covid-19 vaccine, established by the Gamaleya National Research Center for Epidemiology and Microbiology and the Russian Direct Investment Fund (RDIF), into a clients arm throughout a post-registration stage trial at the City Clinic #46 in Moscow, Russia, on Wednesday, Sept. 23, 2020.
Bloomberg | Bloomberg | Getty Images
Russia stated Wednesday its coronavirus vaccine is 92% reliable at avoiding individuals from getting Covid-19, based upon interim trial outcomes.
The statement by Russia’s sovereign wealth fund RDIF came 2 days after U.S. pharmaceutical giant Pfizer and its German partner BioNTech stated their vaccine was more than 90% reliable in stage 3 trials.
RDIF stated the early arise from its late-stage stage 3 scientific trial of the vaccine, called “Sputnik V,” revealed that its effectiveness was “based on the 20 confirmed Covid-19 cases split between vaccinated individuals and those who received the placebo.”
“Currently 40,000 volunteers are taking part in double-blind, randomized, placebo-controlled Phase III of Sputnik V clinical trials, out of which over 20,000 have been vaccinated with the first dose of the vaccine and more than 16,000 with both the first and second doses of the vaccine,” a declaration released on the devoted Sputnik V site stated Wednesday.
In August, Russia ended up being the very first nation to sign up and authorize a coronavirus vaccine. The statement triggered issues from some members of the worldwide clinical neighborhood about the speed of the approval — the vaccine had actually not yet begun stage 3 trials at that point — and absence of readily available information supporting Russian declares for the vaccine’s effectiveness and security.
Russia released some information from early phase scientific trials in September and has actually consistently firmly insisted that its vaccine is safe and reliable. On Wednesday, RDIF stated that “were no unexpected adverse events during the trials. Monitoring of the participants is ongoing.”
Observation of research study individuals will continue for 6 months prior to a complete report of the stage 3 scientific trials exists, RDIF stated, however it kept in mind that the interim research study information will be released by the Gamaleya Center group, which established the vaccine, “in one of the leading international peer-reviewed medical journals” without showing when this may be.
Vaccine race
The Pfizer/BioNTech analysis assessed 94 validated Covid-19 infections amongst its trial’s 43,538 individuals. The partners stated the case split in between immunized people and those who got a placebo showed a vaccine effectiveness rate of above 90% at 7 days after the 2nd dosage.
The most current statement from Russia comes a day after Russia’s President Vladimir Putin revealed that the nation was establishing a 3rd vaccine. He likewise stated Moscow was all set to comply with all other nations on vaccines however alerted versus the “politicization” of the procedure.
Russia has itself been implicated of participating in a vaccine race as drugmakers all over the world attempt to establish an efficient defense versus the infection that has actually triggered over 1.2 million deaths worldwide, according to Johns Hopkins University information. Russia has the 5th greatest variety of validated coronavirus infections, with around 1.8 million cases reported to date.