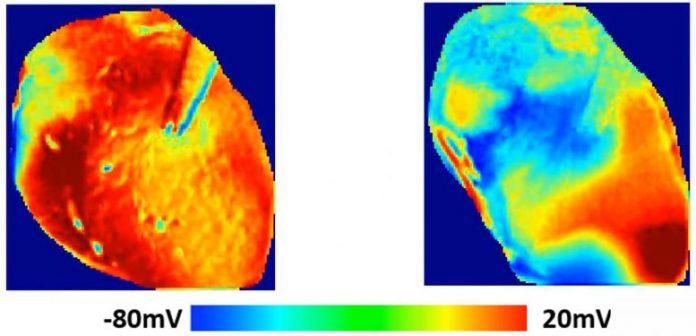
Images reveal the voltage surface area on a bunny heart with and without HCQ. Without the drug (typical) the electrical activation spreads homogeneously, while with HCQ, waves propagate unevenly, creating intricate spatiotemporal patterns and arrhythmias. Credit: Georgia Tech School of Physics
The malaria drug hydroxychloroquine, which has actually been promoted as a prospective treatment for Covid-19, is understood to have possibly major impacts on heart rhythms. Now, a group of scientists has actually utilized an optical mapping system to observe precisely how the drug develops major disruptions in the electrical signals that govern heart beat.
The research study, reported on May 29, 2020, in the journal Heart Rhythm, discovered that the drug made it “surprisingly easy” to set off uneasy arrhythmias in 2 kinds of animal hearts by changing the timing of the electrical waves that manage heart beat. While the findings of animal research studies can’t always be generalized to human beings, the videos developed by the research study group plainly demonstrate how the drug can trigger heart electrical signals to end up being inefficient.
“We have illustrated experimentally how the drug actually changes the waves in the heart, and how that can initiate an arrhythmia,” stated Flavio Fenton, a teacher in the School of Physics at the Georgia Institute of Technology and the paper’s matching author. “We have demonstrated that with optical mapping, which allows us to see exactly how the waveform is changing. This gives us a visual demonstration of how the drug can alter the wave propagation in the heart.”
Images reveal the voltage surface area on a bunny heart with and without HCQ. Without the drug (typical) the electrical activation spreads homogeneously, while with HCQ, waves propagate unevenly, creating intricate spatiotemporal patterns and arrhythmias. Credit: Georgia Tech School of Physics
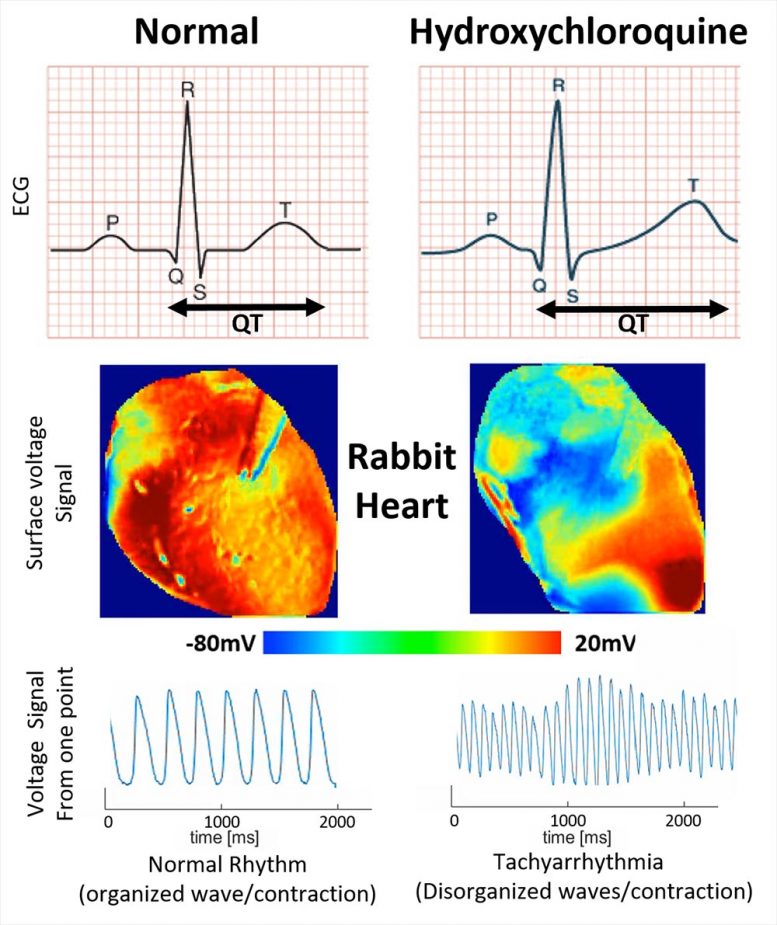
What the group saw was an elongation of the T wave, a part of the heart cycle throughout which voltages generally dissipate in preparation for the next beat. By extending the QT part of one wave cycle, the drug sets the phase for disruptions in the next wave, possibly developing an arrhythmia. Such disruptions can shift to fibrillation that hinders the heart’s capability to pump.
The capability to quickly set off disruptions called “long QT” enhances warns about utilizing hydroxychloroquine (HCQ) in human beings — especially in those who might have heart damage from Covid-19, warned Dr. Shahriar Iravanian, a co-author of the paper and a cardiologist in the Division of Cardiology, Section of Electrophysiology, at Emory University Hospital.
“The hearts used in the study are small and very resistant to this form of arrhythmia,” Iravanian stated. “If we had not seen any HCQ-induced arrhythmias in this model, the results would not have been reassuring. However, in reality, we observed that HCQ readily induced arrhythmia in those hearts. This finding is very concerning and, in combination with the clinical reports of sudden death and arrhythmia in Covid-19 patients taking HCQ, suggests that the drug should be considered a potentially harmful medication and its use in Covid-19 patients be restricted to clinical trial settings.”
Georgia Tech postdoctoral fellow Ilija Uzelac administered HCQ to the animal hearts — one from a guinea pig and one from a bunny — while measuring wave patterns altering throughout the hearts utilizing a high-powered, LED-based optical mapping system. Voltage-delicate fluorescent dyes made the electrical waves noticeable as they crossed the surface area of the hearts.
“The effect of the arrhythmia and the long QT was quite obvious,” stated Uzelac. “HCQ shifts the wavelengths to larger values, and when we quantified the dispersion of the electrical current in portions of the heart, we saw the extension of the voltage across the tissue. The change was very dramatic comparing the waveforms in the heart with and without the HCQ.”
The drug concentration utilized in the research study was at the luxury of what’s being advised for human beings. HCQ generally takes a couple of days to collect in the body, so the scientists utilized a greater preliminary dosage to replicate the drug’s impact in time.
In a regular heart beat, an electrical wave is produced in specialized cells of a heart’s best atrium. The wave propagates through the whole atria and after that to the ventricles. As the wave moves through the heart, the electrical prospective developed causes calcium ions to be launched, which promotes contraction of the heart muscle in a collaborated pattern.
Drugs such as HCQ customize the residential or commercial properties of these ion channels and prevent the circulation of potassium currents, which extends the length of the electrical waves and develops spatial variations in their residential or commercial properties. Ultimately, that can cause the advancement of precariously quick and inefficient heart rhythms.
“The wavelength becomes less homogeneous and that affects the propagation of additional waves, producing sections of the heart where the waves do not propagate well,” Fenton stated. “In the worst case, there are multiple waves going in different directions. Every section of the heart is contracting at a different time, so the heart is just quivering. At that point, it can no longer pump blood throughout the body.”
Patients taking HCQ for illness such as lupus and rheumatoid arthritis hardly ever struggle with arrythmia due to the fact that the dosages they take are smaller sized than those being advised for Covid-19 clients, Iravanian stated.
“Covid-19 patients are different and are at a much higher risk of HCQ-induced arrhythmia,” he stated. “Not only is the proposed dose of HCQ for Covid-19 patients two to three times the usual dose, but Covid-19 has effects on the heart and lowers potassium levels, further increasing the risk of arrythmias.”
Fenton and his coworkers have actually currently started a brand-new research study to examine the impacts of HCQ with the antibiotic azithromycin, which has actually been recommended as a buddy treatment. Azithromycin can likewise trigger the long QT impact, possibly increasing the influence on Covid-19 clients.
###
Reference: “Fatal arrhythmias: Another reason medical professionals stay careful about chloroquine/hydroxychloroquine for dealing with COVID-19” by Ilija Uzelac, Ph.D., Shahriar Iravanian, MD, Ph.D., Hiroshi Ashikaga, MD, Ph.D, NealK. Bhatia, MD, Conner Herndon, MS, Abouzar Kaboudian, Ph.D., James C. Gumbart,Ph.D., Elizabeth M. Cherry, Ph.D., Flavio H. Fenton, Ph.D., 29 May 2020, Heart Rhythm.
DOI: 10.1016/j.hrthm.2020.05.030
The research study, which was supported by grants from the National Institutes of Health and National Science Foundation, was likewise coauthored by Dr. Hiroshi Ashikaga from Johns Hopkins University School of Medicine; Dr. Neal Bathia from the Division of Cardiology, Section of Electrophysiology at Emory University Hospital; Conner Herndon, Abouzar Kaboudian, and James C. Gumbart from the Georgia Tech School of Physics, and Elizabeth Cherry from the Georgia Tech School of Computational Science and Engineering.
This research study is supported by the National Institutes of Health under award number 1R01HL143450-01 and the National Science Foundation under grant 1446675. The material is exclusively the obligation of the authors and does not always represent the main views of the National Institutes of Health or the National Science Foundation.